Research Articles
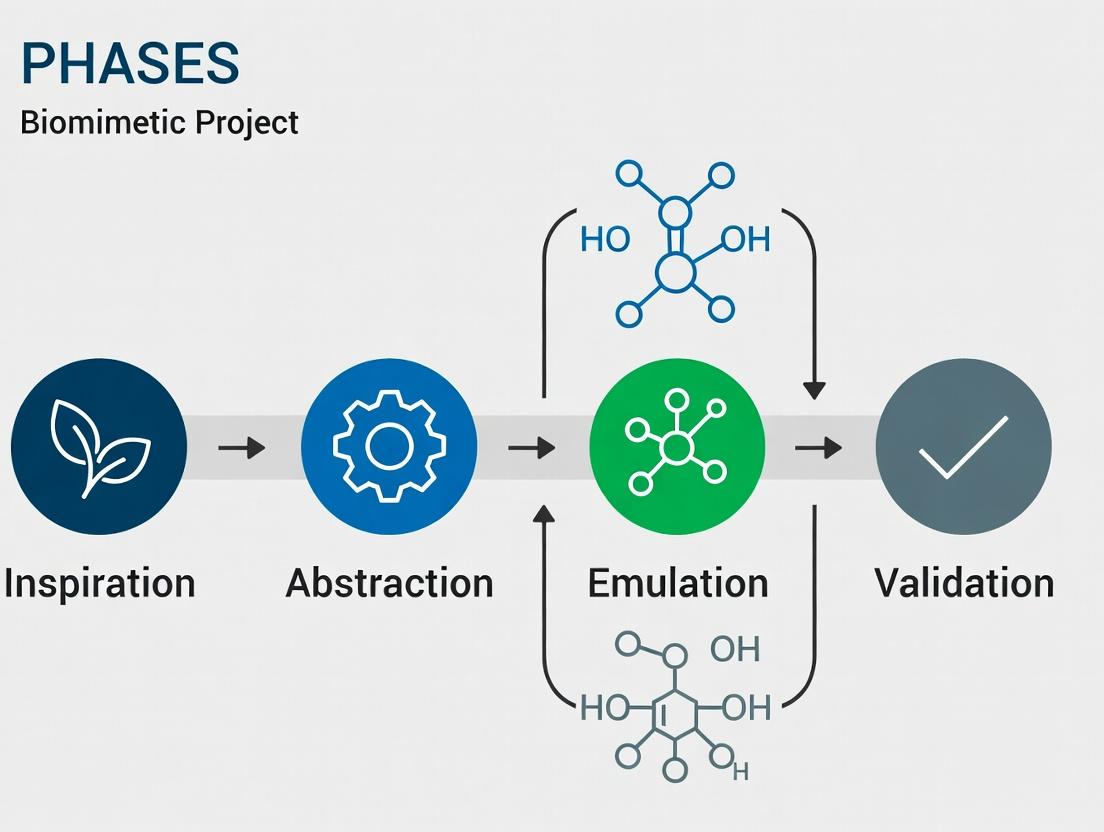
A Sustainable Blueprint: Integrating Eco-Design Principles Across Biomimetic Drug Development Phases
This article provides a comprehensive framework for embedding sustainability into the lifecycle of biomimetic biomedical projects.
Structural Integrity of Extracellular Vesicles: A Comprehensive Guide for Research and Therapeutic Applications
This article provides a thorough examination of extracellular vesicle (EV) structural stability, addressing critical concerns for researchers and drug development professionals.
Overcoming Stability Challenges in Carrier-Free Nanomedicines: Strategies for Robust Drug Formulation
Carrier-free nanomedicines (CFNs) offer a paradigm shift in drug delivery by eliminating exogenous carriers, promising higher drug loading, simpler composition, and improved biocompatibility.
Liposome Stability and Shelf-Life: 2024 Challenges and Solutions for Pharmaceutical Development
This comprehensive review addresses the critical stability and shelf-life challenges facing liposomal formulations in pharmaceutical development.
Navigating the Maze: A Comprehensive Guide to Regulatory Hurdles in Collaborative Nanomedicine Development
This article provides researchers, scientists, and drug development professionals with a strategic framework for addressing the complex regulatory landscape of collaborative nanomedicine projects.
Navigating the Regulatory Maze: A Comprehensive Guide to Nanomaterial Medical Device Approval
This article provides a detailed roadmap for researchers and drug development professionals navigating the complex and evolving regulatory landscape for nanomaterial-enhanced medical devices.
Taming the Blink: Advanced Strategies to Suppress Quantum Dot Blinking for High-Fidelity Single-Molecule Tracking in Biomedical Research
This article provides a comprehensive analysis of quantum dot (QD) blinking, a fundamental photophysical phenomenon that limits their utility in single-molecule tracking (SMT).
Navigating Power Asymmetries in Biomimetic Research: Strategies for Equitable Collaboration in Drug Development
This article examines the complex power dynamics inherent in interdisciplinary biomimetic collaborations between biologists, engineers, chemists, and clinicians.
Beyond Imitation: Deconstructing the Naturalistic Fallacy in Biomimetic Drug Design and Development
This article critically examines the pervasive naturalistic fallacy in biomimetic design for pharmaceutical research.
Nanoparticle Complement Activation: Mechanisms, Mitigation Strategies, and Clinical Translation Challenges
This article provides a comprehensive analysis of nanoparticle-induced complement activation (NICA), a critical barrier in nanomedicine development.